How do you identify methanol?
 Ava White
Ava White 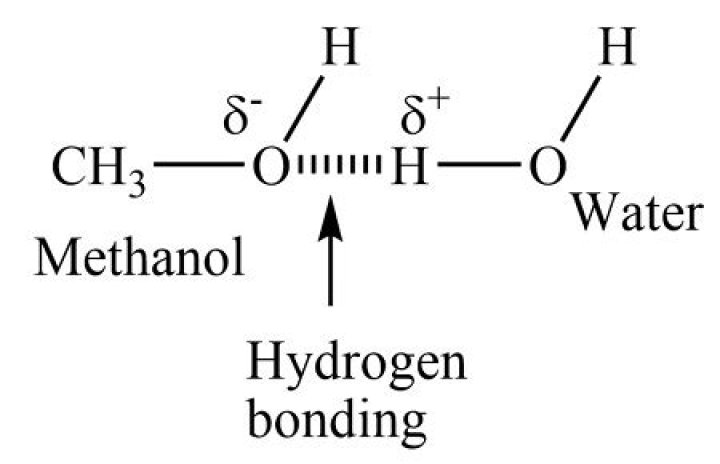
Methanol can be detected by the oil of wintergreen test. In this test, 1-2 ml of methanol along with few crystals of salicylic acid are treated with concentrated sulphuric acid in a test tube which is then gently heated.
oil of wintergreen
Methyl salicylate (oil of wintergreen or wintergreen oil) is an organic compound with the formula C8H8O3. It is the methyl ester of salicylic acid. It is a colorless, viscous liquid with a sweet, fruity odor reminiscent of root beer, but often associatively called "minty", as it is an ingredient in mint candies.
› wiki › Methyl_salicylate
How do I know if I have ethanol or methanol?
Ethanol has a heavy, burning smell and emits bright blue flame. Methanol is unpredictable and has a characteristic odour. When burning it gives off light white flame. Ethanol is typically prepared by the fermentation of food crops from factories.How can you tell methanol from alcohol?
Swirl this container gently a few times, then waft the air from the mouth of the container towards your nose by fanning the air toward you with a hand, with the container placed roughly 8-12 inches from your face. Take note of the scent: If it is pungent and irritating, methanol is present in the alcohol.How do you test methanol in alcohol at home?
To test for the presence of methanol, you can apply sodium dichromate to a sample of the solution. To do so, mix 8 mL of a sodium dichromate solution with 4 mL of sulfuric acid. Swirl gently to mix, then add 10 drops of the mixed solution to a test tube or other small container containing the alcohol.Does methanol look like water?
Methanol is a colourless liquid that boils at 64.96 °C (148.93 °F) and solidifies at −93.9 °C (−137 °F). It forms explosive mixtures with air and burns with a nonluminous flame. It is completely miscible in water.Ethanol or Methanol
What does methanol smell like?
Methanol is a clear, colourless liquid with a faint odor like alcohol. The smell is not very strong and is considered a poor indicator of vapor concentration.What color is methanol flame?
Pure methanol and ethanol generally burn cleanly with a bluish flame color and very limited luminosity. In daylight, it may be difficult to spot such flames.Can you taste methanol?
Methanol is a highly toxic alcohol resembling ethanol in smell and taste. Methanol poisoning is a lethal form of poisoning that can cause severe metabolic acidosis, visual disturbances, and neurological deficit.Can you see methanol burn?
Methanol burns with a clear flame and without smoke, so it is difficult to see in daylight. Methanol will still burn at four parts of water to one part of methanol. Methanol is biodegradable in low concentrations.What is methanol used for illegally?
Abstract. Introduction: Illegal alcohol production remains as a common issue worldwide. Methanol poisoning mostly occurs because of the methanol used in production of counterfeit alcohol instead of ethyl alcohol due to its low price or by drinking the liquids containing methyl alcohol.Is isopropyl alcohol the same as methanol?
Methanol and isopropyl alcohol both have industrial uses, and both are toxic to humans and other mammals. Their chemical structures and other properties differ in several ways. These compounds are not the same.How does methanol taste like?
You can't see, smell or taste it, but drink a small amount and it could make you very ill or even kill you. It's a substance called methanol and it's recently been linked to a number of deaths in Australia and overseas.How do you test for liquid alcohol?
First, the liquid is poured into a tall container, often a graduated cylinder, and then the alcoholmeter/hydrometer is gently lowered into the liquid until it floats freely. Alcoholmeter is used to determine the volume of alcohol or proof.How do you neutralize methanol?
ANTIDOTE: Fomepizole and ethanol are effective antidotes against methanol toxicity. Fomepizole or ethanol should be administered as soon as possible once the patient/victim has been admitted to a medical care facility.How can you tell the difference between methanol and fire?
Methanol flames are almost invisible in bright sunlight conditions, but they may be detected by the heat generated or the burning of other materials. Large amounts of water will remove heat and can be effective in diluting methanol to the point where most fires can be readily extinguished.What happens when methanol is burned?
Like many hydrocarbon derivatives, methanol undergoes combustion (see simulation at bottom of page) when combined with heat and oxygen. This reaction releases energy, carbon dioxide, and water.Do ethanol and methanol smell the same?
Properties of ethanol and methanolMethanol, similarly, is volatile, colourless, flammable liquid with a distinctive smell similar to that of ethanol.



